Abstract
Background: Neurodegeneration with Brain Iron Accumulation (NBIA) is a group of rare genetic disorders characterized by progressive degenerative motor symptoms and the accumulation of iron in the basal ganglia. Pantothenate Kinase-Associated Neurodegeneration (PKAN) is a form of NBIA caused by a mutation in the PANK2 gene leading to a deficiency in pantothenate kinase. Phospholipase A2G6-Associated Neurodegeneration (PLAN) is caused by a mutation in the PLA2G6 gene resulting in impaired phospholipase activity. Current understanding of systemic changes in NBIA disorders is limited, leaving no clear diagnostic biomarkers. Monitoring the systemic changes could identify candidate biomarkers for assessing disease severity and evaluating the efficacy of new therapies.
Previous studies of Parkinson's disease (PD) have found a systemic burden of increased oxidative stress and chronic inflammation accompanies the neurological symptoms of the disease. Similarly, abnormal systemic iron regulation associated with brain iron accumulation as well as damage associated with neuromuscular degeneration could lead to increased oxidative stress and a state of chronic inflammation in NBIA. Our initial investigation of a patient with PLAN1, revealed elevated levels of systemic oxidative stress. We investigated a group of PKAN patients as well as continued our investigation of a patient with PLAN to evaluate the possibility of abnormal iron trafficking, increased oxidative stress and chronic inflammation in NBIA. Our aim was to expand our investigation of circulating levels of inflammatory cytokines, oxidative stress markers and iron regulatory and metabolic proteins in NBIA patients to include a group of patients with PKAN.
Methods: Plasma samples from 15 PKAN patients were collected at the UCSF Benioff Children's Hospital in Oakland, California. Similarly, a plasma sample from a patient with PLAN was collected in Campbell River, British Columbia. Plasma samples from a matched group of 15 healthy controls were also collected at the University of Victoria. All patients provided informed consent to the study.
The pro-inflammatory cytokines IL-6 and TNFα as well as the anti-inflammatory cytokine IL-10 were measured by ELISA. Total levels of the lipid peroxidation product malondialdehyde (MDA) were measured using N-Methyl-Phenyl-Indole (NMPI). Free, acutely generated, MDA not bound to proteins, was measured by removing plasma proteins via a 10KD spin filtration then measuring the MDA content of resulting filtrate using NMPI.
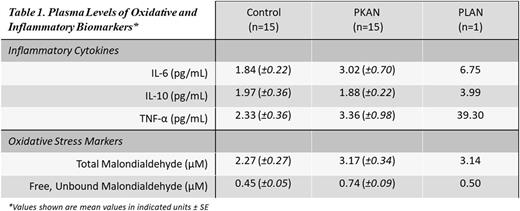
Results: The levels of MDA and Free MDA were significantly elevated in PKAN patients at baseline in comparison to controls (p = 0.05, p = 0.03). IL-6 and TNFα were slightly, but not significantly elevated at baseline compared to controls. We previously demonstrated, similar elevations of oxidative stress in our case study of an NBIA patient with PLAN1. Additionally, all three inflammatory cytokines measured for this study expansion in PLAN were higher than average levels observed in the PKAN and control groups (S ee Table 1). Further analysis of systemic biomarkers in NBIA including proteomic analysis of 30 systemic blood proteins, including iron trafficking proteins is ongoing.
Conclusions: We expand previous findings of elevated levels of systemic oxidative stress in other neurodegenerative diseases such as PD to include NBIA patients with PKAN and PLAN. We provide novel evidence of elevated levels of Free MDA; representative of an acute oxidative stress burden in NBIA in addition to the previously noted elevation in total MDA levels. We provide preliminary signs that of an accompanying inflammatory burden in NBIA, but a larger sample group may be needed to determine its significance.
References
M. Minkley, A. Jackson, D. Smith, C. Borchers, E. Vichinsky, R. Nashmi, P.B. Walter and P. M. Macloed. (2017). Neurodegeneration with Brain Iron Accumulation: PLA2G6-Associated Dystonia-Parkinsonism: Clinical and Animal Studies. Presented at the 2017 European Human Genetics Conference, Copenhagen, Denmark .
Minkley: Apopharma: Research Funding. Neumayr: Apopharma: Research Funding. Walter: Apopharma: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal